|
When primary alcohols oxidize, they make aldehydes. Aldehydes have a carbonyl group (Carbon+Hydrogen+Oxygen) on one end. Common aldehydes in aromatherapy include cinnamaldehyde, citral, citronellal, neral, geranial, myrtenal, and octenal.
Aldehydes tend to be cooling, calming, and uplifting. Their sedative and antispasmodic properties can help you relax and improve your mood. They are great in diffusers because, with their antibacterial and antiviral properties, they can get rid of cold and flu germs that are floating around. Yet, rather than smelling medicinal, they have fresh, herbaceous, floral, or fruity aromas. Those wonderful aromas are the reason you'll even find aldehydes in perfume. There are some cautions that go with oils high in aldehydes. They may irritate the skin and mucous membranes, so they need to be limited to 1% or less of a blend. When used topically, they should be used short term only (meaning a few days at most). Nature derives ethers from phenols. Because of this, you may hear the terms 'phenolic ethers' or 'phenolic methyl ether'. All three terms are referring to the same chemical family. Like phenols, ethers have powerful therapeutic properties and are very harsh on the skin. They also come with significant toxicity warnings. You might ask "If they're so rough on our skin, why use them?".
We use them because, therapeutically, ethers tend to be strongly antibacterial, antifungal, analgesic, anti-inflammatory, antispasmodic, and carminative. They can also provide a local anesthetic effect. Oils rich in ethers include Anise, Fennel, Nutmeg, and Tarragon. They can be used to calm the stomach, relieve gas and bloating, calm muscle spasms, calm cramping, aid the lungs, calm a spastic cough, and promote relaxation so you can sleep. Ether-rich oils are an example of why knowing and understanding the chemistry of essential oils is critical. While they can be highly effective in blends, they must be used in very small quantities and for very short periods of time. Warnings: Ethers can burn the skin and/or cause skin cell damage. There are specific warnings for different ethers listed below. (E)-anethole - Avoid during pregnancy. Do not use if on blood-thinners or if you have a bleeding disorder, or before or after surgery. It has possible estrogenic activity, so do not use if you have or have had estrogen related conditions like endometriosis or estrogen-related cancers. Elemicin: Some studies on male mice indicated a weak possibility of being carcinogenic, but there is not enough evidence to make an actual determination of this. Estragole (aka: methyl chavicol): Do not use if you are taking blood-thinners or if you have a bleeding disorder, before or after surgery, or if you have a peptic ulcer. Myristicin: no warnings when used properly in aromatherapy, Personal note: There are so many essential oils available that I find I rarely see the need to use ether-rich oils. Often, I will use the herb instead of the oil -- and that for limited time too. Datasheets for these essential oils will be coming soon in the membership section. Phenols are very powerful chemicals! In essential oils, structurally, they seem similar to alcohols, but have a phenyl or benzene ring and have one or more hydrogen atom(s) that get swapped out with another group of atoms. The phenols we find in essential oils are carvacrol, cresol, eugenol, and thymol. You'll notice that they all have the -ol- suffix like the alcohols (monoterpenols and sesquiterpenols).
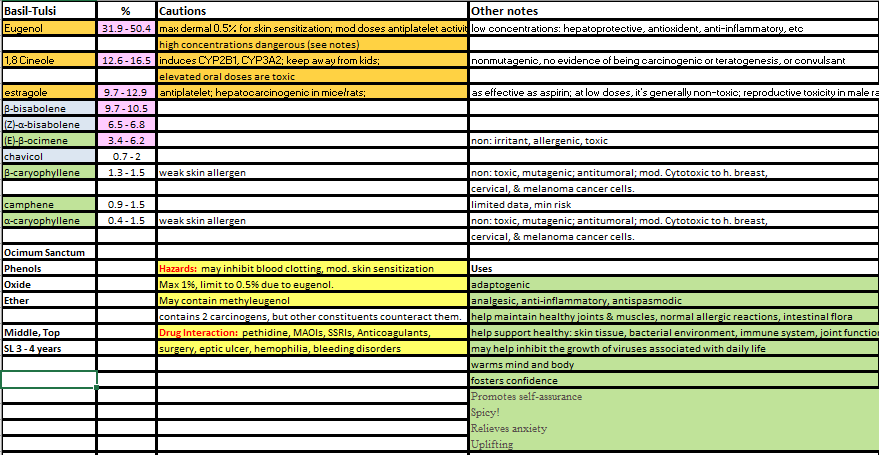
These chemicals are strongly antiseptic, antibacterial, antifungal, antimicrobial, germicidal, bactericidal, and immune-system boosters. When you're sick, they're great in a diffuser to help knock germs out of the air and to help your immune system fight off illnesses. They are also CNS stimulants and can be of assistance with certain types of depression. Eugenol, in small amounts, can be beneficial to the liver; however, in larger amounts it becomes toxic to the liver. Essential oils that have significant levels of phenols include Aniseed, Tulsi Basil, Cinnamon, Clove, Oregano, Tarragon, and Thyme ct. thymol. The phenols in these oils are highly reactive and will irritate (possibly damage) the skin and mucous membranes. If you use any of these oils topically, it's important to dilute them very well in a carrier oil that is skin healing. A 0.5% maximum dilution is recommended -- that's 3 drops EO per 1 ounce of carrier oil. When diffusing, limit the total phenol content to a maximum of 10% of the diffuser blend. i.e. for a 5 mL diffuser blend, phenol content should be 1/2 mL or less. Additionally, such blends should be used short-term -- for only two or three days. Other precautions to follow when using oils high in phenols include: * Do not use if you have a blood-clotting disorder or are taking blood thinners. * Do not use before surgery. * Do not use in a bath. * Do not use on babies or children. * Do not use if you have 'hot' skin conditions (i.e. dermatitis, eczema, psoriasis) * Do not use on broken or irritated skin. * Be sure to look at your GC/MS reports and calculate out your phenol percentages when blending essential oils. Below are samples of a couple of spreadsheets I made when I first started using essential oils (and before I got certified as an aromatherapist). The first is for a single oil -- Tulsi Basil. The second is part of a blend where I calculated percentages of chemicals within the blend and compared them to make sure I didn't use too many harsh constituents. Making these charts helps me visualize my blends as a whole and ensure I keep them safe. Oxides are cyclic ethers that are usually made from a monoterpenols or sesquiterpenols (alcohols). In most cases, the word 'oxide' is added to the monoterpenol or sesquiterpenol name. The oxides found in Essential Oils are Bisabolol oxide, Bisabolone oxide, Caryophyllene oxide, Linalool oxide, Pinene oxide, Sclareol oxide, and 1,8-Cineole. [You'll notice the 1,8-Cineole (aka Cineole) doesn't use the word 'oxide' -- it's an exception to the rule.]
Oils with high levels of oxides are, perhaps, best known for breathing. They break up excess mucus in the sinuses and lungs and help get rid of it (decongestant and expectorant). You've probably already experienced this effect with the 1,8-Cineole in Eucalyptus. If you've ever had a cold or the flu, you may have used an OTC product containing Eucalyptus and gotten temporary relief from congestion and been able to breathe more easily. You may have even recovered a bit more quickly, because oxides generally have antibacterial and antiviral properties. Did you know that essential oils that are oxide-rich can also help you focus and be more alert? This effect goes hand-in-hand with breathing. These oils may help improve circulation which means more blood and oxygen are available to your brain when you inhale them. There are some precautions to take into account when using essential oils with oxide content. Do not use these oils with children under the age of 10, or on or near their faces -- oxides can slow down the central nervous system and/or breathing in young children. Do not use if you are allergic to oxides. If you have asthma, test a very small amount first. While they may help open the airways in the presence of asthma, they have the potential to exacerbate the condition. When using oxide-rich oils topically, limit them to a 2% maximum dilution to avoid potential skin irritation. Esters occur when acid and alcohol molecules bond together. This bond creates chemicals that are gentle on the skin and have balancing energetics. Aromatically, they are often flowery and/or fruity middle notes, and their shelf life is usually 3 - 5 years.
Essential oils that are ester-rich are generally soothing, uplifting, and sedative. They can help balance your body during times of high stress. Several can relieve pain and inflammation. They are great on skin irritations like bites, stings, rashes, and skin conditions, and some can even help improve digestion. Overall, essential oils high in esters are safe to use, but there are some precautions that need to be taken into consideration. * The acid-alcohol bonds can be broken if the essential oil is stored improperly (i.e. in a hot, humid area like a bathroom), therefore, store these oils in a cool, dry area. * Wintergreen and Birch Essential Oils have an ester - methyl salicylate - in very high levels. [This chemical is the plant remedy used before aspirin, and is the inspiration behind the creation of aspirin (aspirin = acetyl salicylate).] These oils need to be used by adults only and in low dilutions for limited time periods. This chemical constituent is not easily processed by the human liver and can reach toxic levels if used improperly. See the datasheets in the membership section for more information. If you are allergic to aspirin, do not use Wintergreen or Birch essential oils. Like monoterpenols, sesquiterpenols are alcohols and have the -ol- suffix. They have a backbone of 15 carbon atoms (instead of 10) and a hydroxyl group (an oxygen-hydrogen pair) attached to the molecule. This has an effect on their therapeutic properties, size, and longevity.
Because they are alcohols, sesquiterpenes tend to be antibacterial, immune stimulants, cooling, antispasmodic, and sedative. Oils high in sesquiterpenes are good in deodorants, lotions, blends for skin irritations, and diffuser blends for sleep or to ease breathing. The 15 carbon backbone makes the molecules larger and heavier. These larger molecules deepen the aroma to middle-base and base notes which last longer (as opposed to evaporating quickly). These deeper notes offer grounding and calming energetics to a blend. The larger molecules also absorb more slowly, which means they have a longer time to impart their therapeutic properties to the skin, and they give a longer shelf life to essential oils rich in these chemicals - about 6 - 8 years (or longer). While sesquiterpenes are found in many essential oils, only a few oils are sesquiterpenol-rich. They include Cedarwood, Myrrh, Patchouli, Sandalwood, and Vetiver. Similar to monoterpenes, sesquiterpenes are terpenes, which means the molecules consist of only carbon and hydrogen atoms. (By now, you'll notice that terpenes have an -ene- suffix.) These molecules are larger and heavier than monoterpenes because they have a skeleton of 15 carbons. As a result, they evaporate more slowly and tend to have deeper, richer, more enduring aromas. A few of my favorites are Beta-caryophyllene, Alpha-humulene, Zingiberene, and Alpha-curcumene.
While there are some common therapeutic properties in oils that have high levels of sesquiterpenes, there are differences as well. Those common properties include grounding, antiseptic, antibacterial, analgesic, anti-inflammatory, and antispasmodic. However, in this category, individual oils must be studied for their positive effects because some are sedative, some can help lower blood pressure, some are skin-healing, and some are cooling while others are warming. Many sesquiterpene-rich oils are gentle on the skin, but some can irritate the skin if they have oxidized. Monoterpenols are a family of chemicals with 10 carbon molecules that have a functional oxygen-hydrogen group attached. (I know, chemistry -- or, yay! chemistry!) The -ol- suffix indicates that monoterpenols are alcohols. Most of the chemicals in this family have the -ol- suffix: i.e. Geraniol, Linalool, Citronellol, and Menthol. Consider the effects that small to moderate amounts of alcohol have on the body -- try to list some. See how many of yours match mine.
Relaxing, disinfecting, sedative, calming, antispasmodic, antianxiety, cooling All of these are therapeutic properties found in monoterpenols. It gets better though, because monoterpenols have even more benefits. The majority tend to be anti-inflammatory, antifungal, antiviral, deodorizing, most are skin-friendly, some are neuroprotective, and many stimulate the immune system. While most monoterpenol-rich essential oils are safe, those high in geraniol may be mild skin sensitizers, so the recommended maximum level for dermal application is 5.3%. There is one monoterpenol that is very popular, but comes with precautions -- Menthol. We find a high level of menthol in Cornmint and Peppermint essential oils. Rather than being sedating, menthol is stimulating - it'll wake you up and help you focus. Applied topically, its cooling sensation makes a good analgesic, but it can be harsh on skin and mucous membrane cells, so it's best used in smaller dilutions. More significantly, it needs to be kept away from the faces of children from newborn to age 5 as it can cause breathing problems. People with G6PD deficiency need to avoid menthol because their bodies cannot metabolize it. A chemical family refers to a set of chemicals that have specific traits in common in their molecular structure. This similarity in molecular structure means the chemicals will usually have similar therapeutic properties and precautions.
Molecules in the monoterpene chemical family contain only 10 carbon atoms and a varying number of hydrogen atoms. Chemicals within this family include (but are not limited to) alpha-pinene, beta-pinene, camphene, limonene, phellandrene, terpinene, and terpinolene. Many essential oils with high levels of monoterpenes tend to be antibacterial and antiviral. As such, they are good for diffusing during cold and flu season and for using in soapmaking. Monoterpenes in general are also anti-inflammatory. Some have a warming effect which can help soothe pain. This makes these appropriate choices for after-exercise topical blends. Energetically, oils rich in monoterpenes can lift the spirit and encourage energy. Think about citrus oils like lemon, orange, and lime -- these are monoterpene-rich oils. How do they make you feel? Happy? Full of energy? When using monoterpene-rich essential oils, it's important to store them properly (in a cool, dark space) as they oxidize (spoil) easily. When oxidized, they may irritate the skin or cause sensitization. Did you know that all of the nature-made chemicals found in essential oils can be categorized in chemical families? These families are Monoterpenes, Monoterpenols, Sesquiterpenes, Sesquiterpenols, Esters, Oxides, Phenols, Aldehydes, Lactones, Ketones, Coumarins, and Ethers. The structure of the molecules of each chemical determines the family to which it belongs. The families may have certain, generalized, therapeutic property tendencies, and/or certain, generalized cautions.
An average oil may have around 100 chemicals. When we list those chemicals by family, we will usually see that the oil contains a higher percentage of one or two families. For example, an oil with 54% of monoterpenes would be considered monoterpene-rich, and its therapeutic properties would be consistent with those of the monoterpene family. |
DisclaimerThe information contained in this blog is for educational purposes only and has not been evaluated by the Food and Drug Administration. Archives
February 2021
Categories |
 RSS Feed
RSS Feed